After your patients with PSMA+ mCRPC receive their first ARPI, be ready for what's next.1 Be ready to rethink the sequence.*
*For patients considered appropriate to delay taxane-based chemotherapy.1
Identifying Patients
1 ARPI could have been received at any point in your patient’s prostate cancer journey, including in the castration-sensitive setting1,2
For your patients on an ARPI, at what point do you begin considering subsequent treatment options?
Patient Profiles
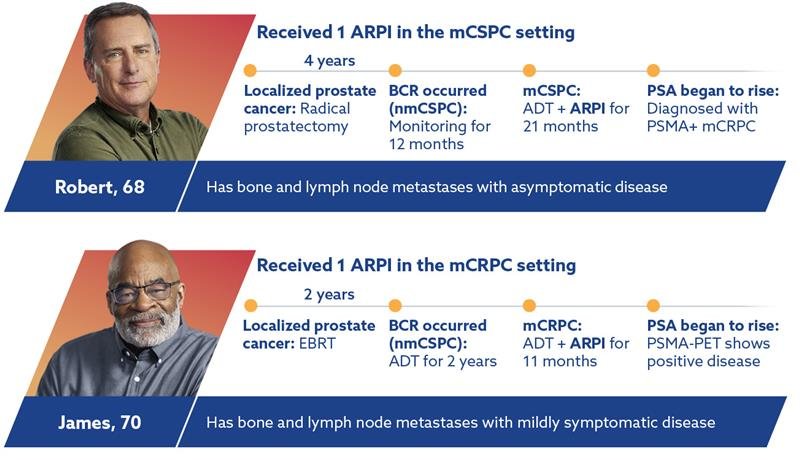
MEET PATIENTS WITH PSMA+ mCRPC WHO ARE ELIGIBLE FOR PLUVICTO
Plan for PLUVICTO after only 1 ARPI, which could have been received at any point in your patient’s journey1,2
Hypothetical patient cases.
ADT, androgen deprivation therapy; BCR, biochemical recurrence; EBRT, external beam radiation therapy; mCSPC, metastatic castration-sensitive prostate cancer; nmCSPC, nonmetastatic castration-sensitive prostate cancer; PET, positron emission tomography; PSA, prostate-specific antigen; PSMA, prostate-specific membrane antigen.
Advancing PLUVICTO Therapy for the Right Patients
How to Communicate PLUVICTO Radiation Safety With Your Patients
Plan for PLUVICTO
GET YOUR PATIENTS ON PLUVICTO PROMPTLY AFTER ONLY 1 ARPI1
3 ways to accelerate the PLUVICTO referral process
*There are over 765 PLUVICTO treatment sites in the US. Please note that the PLUVICTO Treatment Centers listed are only those that have authorized their participation on the PLUVICTO website.4
†Primary plans only; an additional 1-2 days if secondary plan coverage review is required.
Which of your patients with PSMA+ mCRPC could benefit from PLUVICTO now?